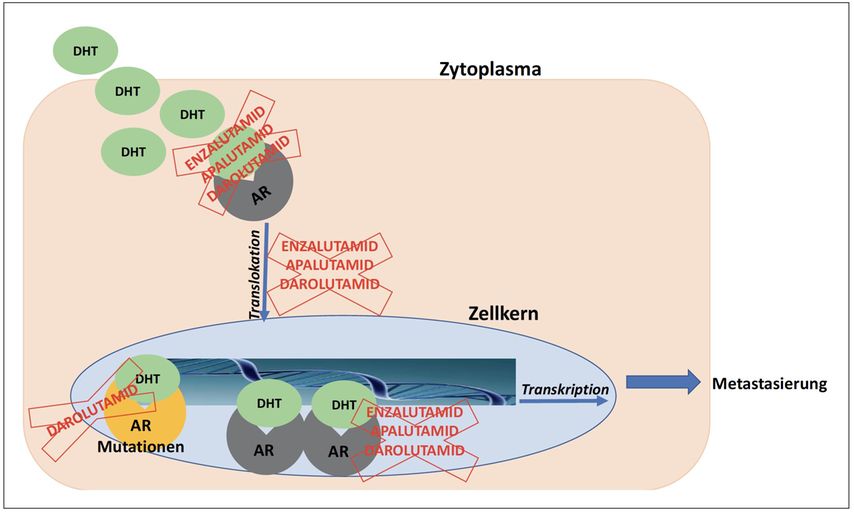
Proxalutamide (developmental code name GT-0918) is a nonsteroidal antiandrogen (NSAA) – specifically, a selective high-affinity silent antagonist of the androgen receptor (AR) – which is under development by Suzhou Kintor Pharmaceuticals, inc., a subsidiary of Kintor Pharmaceutical Limited, for the potential treatment of COVID-19, prostate cancer, and breast cancer. It was approved in Paraguay for the treatment of COVID-19 in July 2021, but has not been approved at this time in other countries.
Research
Prostate cancer
Proxalutamide is in phase III studies for mCRPC as monotherapy and in combination with abiraterone. In the United States, it is in phase II study as monotherapy for mCRPC.
Other indications
Proxalutamide is in phase Ic clinical trial in China.
See also
- List of investigational sex-hormonal agents § Androgenics
References